
Figure 3 from Proline editing: a general and practical approach to the synthesis of functionally and structurally diverse peptides. Analysis of steric versus stereoelectronic effects of 4-substituted prolines on conformation within peptides.

Asymmetric Catalytic Ring‐Expansion of 3‐Methyleneazetidines with α‐Diazo Pyrazoamides towards Proline‐Derivatives - Wang - Angewandte Chemie International Edition - Wiley Online Library

Locked Conformations for Proline Pyrrolidine Ring: Synthesis and Conformational Analysis of cis- and trans-4-tert-Butylprolines | The Journal of Organic Chemistry

Why are glycine and proline less commonly found in alpha helices than other amino acids? | Homework.Study.com
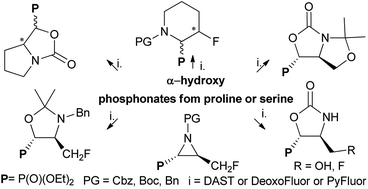
Pyrrolidine and oxazolidine ring transformations in proline and serine derivatives of α-hydroxyphosphonates induced by deoxyfluorinating reagents - RSC Advances (RSC Publishing)

Locked Conformations for Proline Pyrrolidine Ring: Synthesis and Conformational Analysis of cis- and trans-4-tert-Butylprolines | The Journal of Organic Chemistry

Asymmetric Catalytic Ring‐Expansion of 3‐Methyleneazetidines with α‐Diazo Pyrazoamides towards Proline‐Derivatives - Wang - Angewandte Chemie International Edition - Wiley Online Library

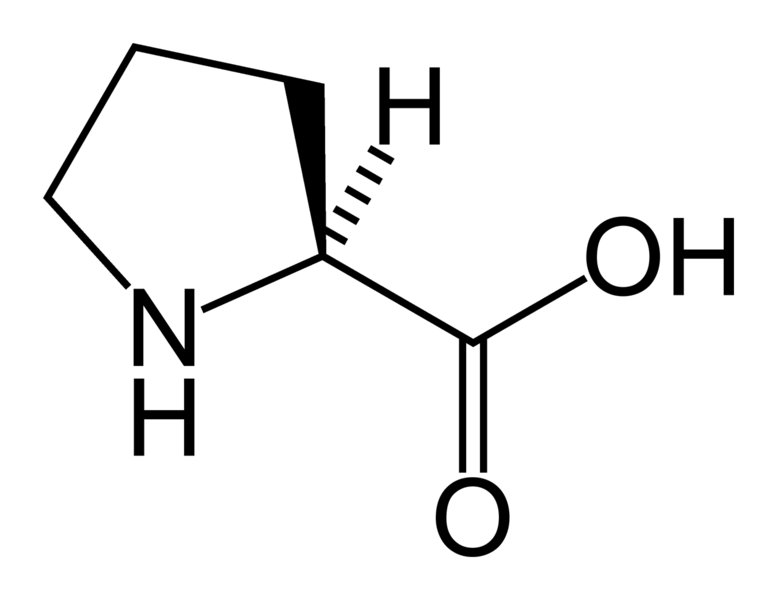







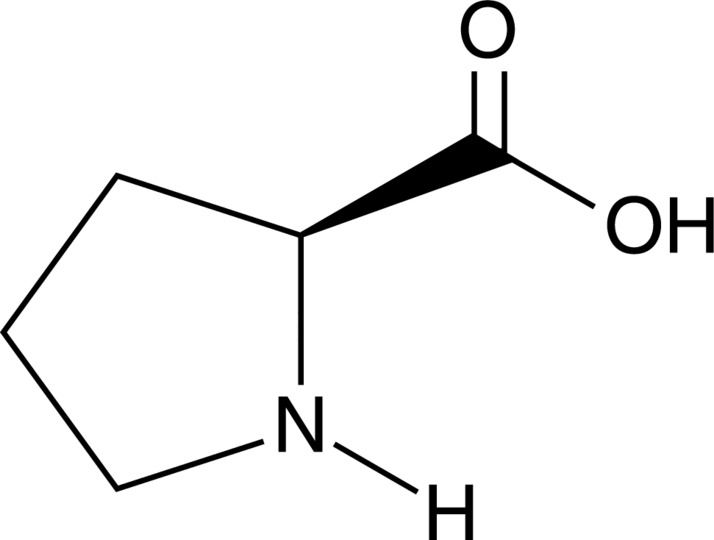



![Q67P After the peptide chain of colla... [FREE SOLUTION] | StudySmarter Q67P After the peptide chain of colla... [FREE SOLUTION] | StudySmarter](https://studysmarter-mediafiles.s3.amazonaws.com/media/textbook-exercise-images/image_VmOU0Uw.png?X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Credential=AKIA4OLDUDE42UZHAIET%2F20230524%2Feu-central-1%2Fs3%2Faws4_request&X-Amz-Date=20230524T035013Z&X-Amz-Expires=90000&X-Amz-SignedHeaders=host&X-Amz-Signature=b04c5e9001790b643d98255c50f3f3115606b96eaf9a9c138c200b8846da8dc1)
